For the past half year, I have been building hardware to remove carbon dioxide from ambient air at the lowest cost ever. This post is the first part of a series that tells the story of how I got here, the fundamentals of direct air capture (DAC), and my approach to solving this problem.
But before we begin:
The Castle is where I reside. If you are interested in fighting humanity’s war against carbon dioxide or if you simply love building hardware, please apply at Terraform Industries or reach out directly.
My Origin Story
For a brief moment in middle school, I get infected by the “global warming is going to end us soon” mind virus. It comes from consuming the following cocktail of ideas in a science class: fossil fuels are finite, burning them is bad for nature, and disaster for mankind is coming soon. I am still fuzzy about what the end of the world would look like, but I am scared-straight after watching the movie “2012”. My ills are cured after observing my parents’ nonchalance about this impending catastrophe and reading the geoengineering section of my science textbook. We might just be fine.
Years later, a different virus descends upon us. COVID never catches me, but it traps me inside. I devote part of my screen time to a MOOC about carbon capture and post Reddit-style insights in our chemistry class’s group chat on interesting lessons. I get glazed for my active participation during a parent-teacher meeting, and this feels rewarding. It is difficult to pinpoint causality, but these experiences play some role in my decision to pursue engineering and focus on climate tech in university.
In college, I start by building electric racecars, which is the ultimate technical learning experience. I think making the future of batteries is my North Star. However, three things coalesce and discourage me from going deeper into climate tech: I take a soul-sucking public policy class on energy, a boring internship at Rivian convinces me that all the low-hanging fruit in the EV industry have been picked, and I get attracted to other shiny problems.
Fast forward to a month after graduation, where I read my onboarding document at Terraform Industries (TI) and realize that I am taking over the Carbonator’s design, a reactor for direct air capture of CO₂. This feels like a crude full-circle moment. For reasons I can’t fully explain, I picture the bubbles in a Coke bottle when I read about the Carbonator. The carbon dioxide in carbonated products turns out to be a potential product line, but before we get to that part of the story, we must go back to the basics.
Fundamentals of Direct Air Capture

A covalent double bond connects the carbon and oxygen atoms. The distance between their nuclei is short, and their similar size enables good electron orbital overlap. These factors combine to make a strong bond that is difficult to break. Life seems hard for the DAC engineer who has to find this fully oxidized and inert molecule that is not abundant in air. Luckily, there is a chink in the armor of carbon dioxide.
Oxygen is super-duper electronegative, and it pulls the electron cloud in the double bond toward itself. This gives the molecule some local polarity, as the carbon is slightly positive and the oxygen is slightly negative. The electron-poor carbon atom can be attacked with a base—a substance that likes to give away its electrons. This acid-base reaction is the most common way that carbon dioxide is captured from air. It happens even under ambient conditions because the products are stabler than the reactants, and it feels like a game about finding ways for molecules to climb down the energy landscape.
For all the talk about rising CO₂ levels, there is only ~0.04% of it in ambient air, which makes DAC akin to finding a needle in the haystack. Physically feasible solutions have been around for close to a century, but the current moonshot for a carbon capture device is going from $1000 / ton to $100 / ton of CO₂ captured.
TI’s plan to achieve this is based on “calcium looping,” and the sketch below shows the cycle that allows us to reuse the calcium-based sorbent to gobble up the CO₂. The hydration step converts quicklime (calcium oxide) to slaked lime (calcium hydroxide), and a flaking machine converts a slurry of this into flakes that remind you of Tony the Tiger. The carbonation step converts these flakes into limestone (calcium carbonate) by blasting a lot of humid air through them. Finally, a very-very-very hot kiln decomposes this carbonate to release CO₂ and calcium oxide, which is fed back into the input of the hydrator to complete the loop.
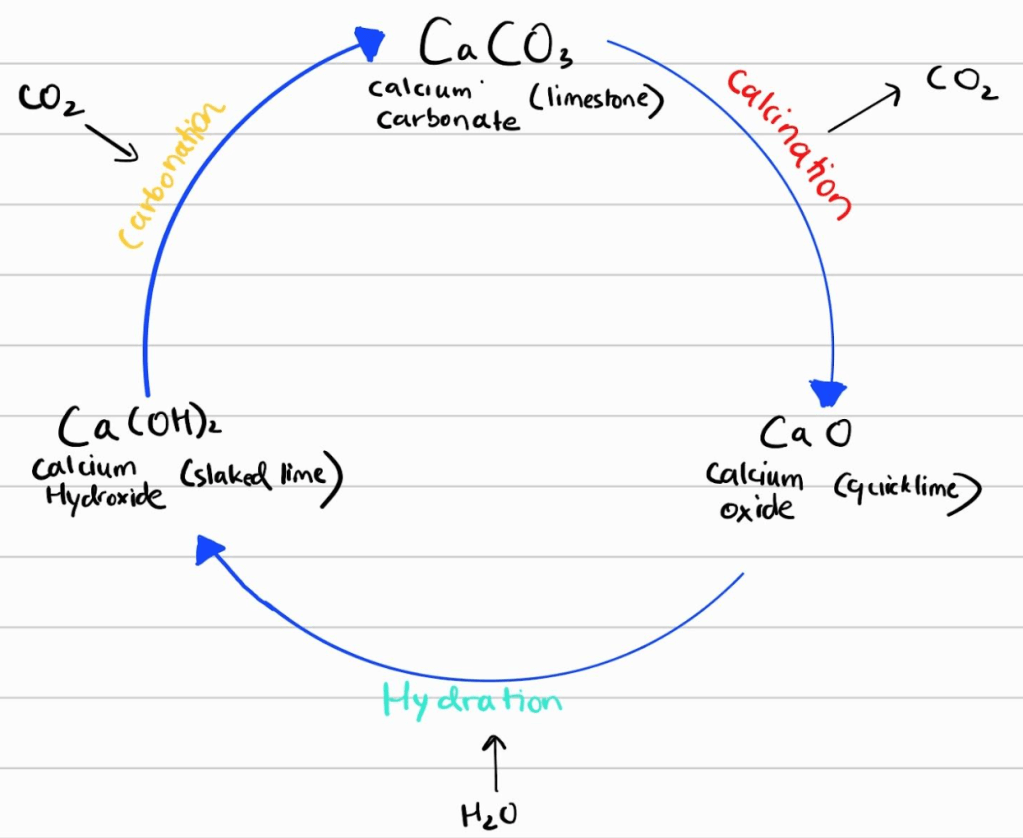
I have focused on the carbonation step, which is thermodynamically feasible at ambient conditions but kinetically infeasible without water. The water acts as the battlefront for the reaction, and it comes from three sources: the humidity in the air, the initial moisture of the flakes, and as a byproduct of the reaction. The initial moisture of the flakes is what holds them together for long enough to prevent disintegration into powder. It also provides the starting burst of carbonation that leads to a shell forming around the flake—think soft and moist flakes turning into crispy frosted flakes. The reaction produces heat, but large amounts of humid air keep the flakes cool and wet, allowing more of the flake to carbonate.
The water is critical because polar H₂O molecules partially break apart the ionic structure of calcium hydroxide, which dissociates into calcium and hydroxide ions. When the CO₂ molecule finds its way to the battlefront, it dissolves and gets attacked by the hydroxide ions, which break the double bond and form a bicarbonate ion, and another hydroxide ion strips the proton off this bicarbonate to form a carbonate ion. Now we have calcium ions and carbonate ions floating around in an aqueous solution that react almost instantaneously and precipitate into solid calcium carbonate.

The reaction itself is really fast; the hard part is getting the CO₂ to reach the aqueous solution of calcium ions. The flake is initially composed of calcium hydroxide and some water, but a shell of calcium carbonate grows on this porous flake until most of the hydroxide is trapped inside. As more carbonate forms, the unreacted calcium hydroxide core shrinks, making it harder for the CO₂ to find the calcium hydroxide, and thus carbonation slows down.
AK’s law of carbonation states that to get a lot of needles, one needs a lot of haystacks. So we use powerful fans that sound like little jet engines to blow 5,000 liters per second of air (you just breathed half a liter of air in the second it took to read the line). Despite getting blasted with all this air, it takes a day for half the flakes to turn into calcium carbonate. This is because the diffusion resistance scales with the amount of carbonate in the flake. Like the diminishing pleasure of guzzling each successive pint of cookies and cream, the returns from blowing more air on heavily carbonated flakes drop off sharply. When the rate has slowed below a cutoff, the Carbonator is ready to dump these flakes into the calcination kiln.
You can do all the chemical analysis in the world to figure out your carbonation conversion, but the Rick Rubin way to FEEL the carbonation progress is to release a handful of flakes and hear the sound of pennies falling into a jar.
The Design Philosophy
To make money and achieve the orders of magnitude reduction in cost, we must churn out reactors at the TERRA scale (technically mega scale, but I couldn’t resist). Instead of performance maxxing like a race car engineer, I now design to minimize the time and money spent building the reactor. “SIMPLIFY DELETE REPEAT” is the mantra of the day, the night, and the weekend, and now it has spread to other parts of my life. As if hearing this mantra in every stand-up was not enough, we were handed “Delete” stickers to put on parts that needed elimination.

An ambitious startup must move as fast as LeBron on a fast break. Prototypes must be churned out rapidly and tested against reality. There are many allotropes of these textbook “easier said than done” ideas, such as Elon’s algorithm or the Skunkwork’s manual and they are not new. However, reminders are useful, and their impact on generations of businesses, from factory lines to contemporary SaaS startups, illustrates the power of these methods.
I guess the repetition works because, like calories in Bryan Johnson’s diet, all my parts must fight to survive. I have only ever heard tales of custom CNC-machined components. They never have a happy ending! This forces me to think real hard about making things the simplest they need to be to ensure the survival of my designs. These precautions are essential to stay safe from the hardware gremlins because each added component exposes more surface area for them to attack.
It has taken a few hardware failures to really digest some of these lessons, and I have a long way to go. The part I am struggling with is determining the optimal number of tokens to devote to thinking about a problem before building prototypes. Only reps will tell if this is an intuition I can develop.
These “reps” are the next part of my story.



